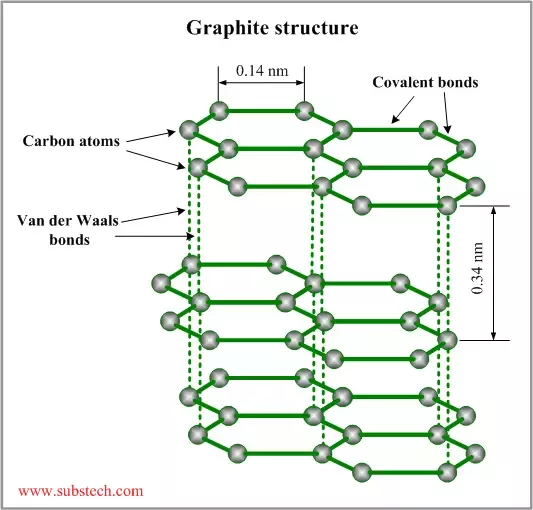
They both have hexagonal and layered structures, in which the covalent-bonded layers are held together by weak van der Waals forces. Due to its high temperature stability, chemical inertness, high thermal conductivity, low thermal expansion and high ion-exchange capabilities, graphite is widely used in many industrial applications 5.
There was much discussion in the comments to the OP regarding whether erasing a pencil mark is a chemical process or merely a mechanical process, akin to removing an oil spill with high pressure water (my analogy). $begingroup$ You can erase the pencil marks with your finger. I'm not convinced by your arguments. One type of eraser absorbs graphite quite heavily, so all erasers function via 'van der waals' interaction? I've mentioned in the comments that this by itself is an extremely vague term. So why do you need to rub your eraser then?
The way I understand it, it is 1) rubbing the eraser thus loosening the graphite 2) some kind of absorption process by the material to prevent smearing, is this correct? $endgroup$–Mar 20 '17 at 9:36.
$begingroup$ Thank you for the answer! The biggest follow-up question I have is what does it mean for a molecule to be 'small'? We say graphene isn't small, but polymer chains are. How do we draw the distinction? If we have a sample of a branched polymer consisting of 100 chains and every 2 chains are joined to each other (so there are 50 pieces), why do we need to break the branch bonds rather than the Van Der Waals forces between the pieces for melting?
That would allow the pieces to move freely. Lastly, aren't Van Der Waals forces weaker than the branch bonds? If so, why aren't these broken first?
$endgroup$–May 1 '17 at 7:41.
